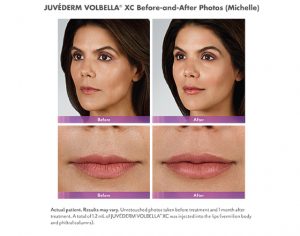
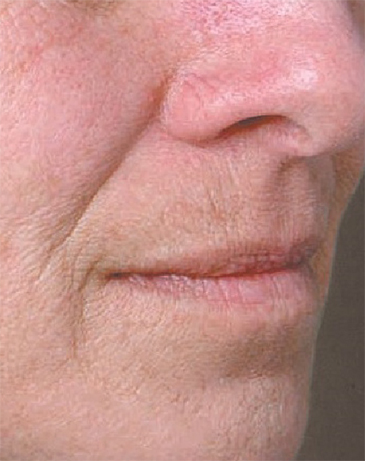
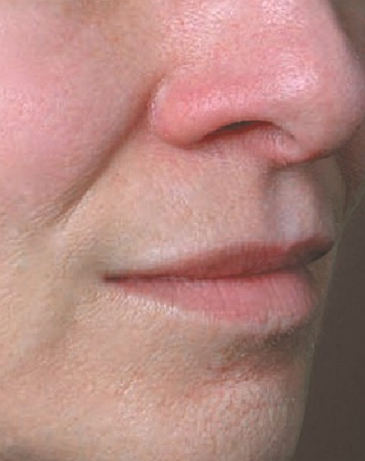
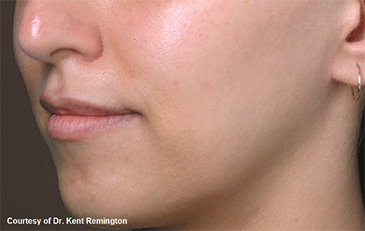
North Naples 239.596.9337 Naples Downtown 239.307.4605 Bonita/Estero 239.498.3376 Fort Myers 239.590.8894
Our offices will be closed Monday, 9/2 in observance of Labor Day. We intend to reopen on Tuesday, 9/3, however, we may be closed due to the hurricane. Please stay tuned to our website for updates.
We are currently experiencing technical difficulties with our phone system. To reach our office, please email us
Our North Naples phone number is being serviced, please call 844.485.0104 to reach a staff member.
Our main phone number for our downtown Naples office is temporarily out of service. If you need to reach us, please call 1-844-485-0104. We apologize for any inconvenience.